TRINTELLIX® (vortioxetine) is indicated for the treatment of Major Depressive Disorder (MDD) in adults.

Patient portrayal.
Individual results may vary.
Safety Data
How the safety profile fits in the story
The safety profile of TRINTELLIX was evaluated in more than 5,800 adult patients across short-term and long-term clinical studies1
Common adverse reactions occurring in ≥2% of patients treated with TRINTELLIX and ≥2% greater than the incidence in placebo-treated patients in 6- to 8-week trials*
Short-term 6- to 8-week studies
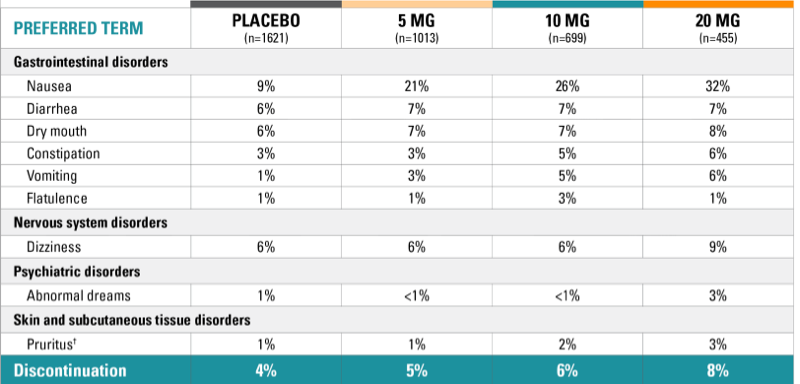
*Based on the rates for 5-, 10-, 15-, and 20-mg doses.
†Includes pruritus generalized.
In the short-term studies, nausea was the most common adverse reaction reported as a reason for discontinuation.

Nausea1
- Nausea was the most common adverse reaction reported as a reason for discontinuation
- Most common adverse reaction; frequency was dose-related
- Intensity: Typically mild or moderate
- Median duration: 2 weeks
- Most commonly occurred in the first week of treatment
- 15% to 20% of patients experienced nausea after 1 to 2 days of treatment
- Remained unresolved for 10% of patients taking TRINTELLIX 10 mg/day to 20 mg/day at the end of the 6- to 8-week placebo-controlled studies

Washington University, Midwest Research Group, St. Charles, Missouri
"I tell patients that nausea, the most common adverse reaction in the short-term studies, typically occurs in the first week of treatment and that 10% of patients taking TRINTELLIX at the 10 mg or 20 mg dose were still experiencing nausea at the end of the 6- to 8-week placebo-controlled studies. The nausea was also dose-related, so I can titrate patients down to a lower dose if it persists."
Dr Mattingly is a physician and principal investigator in clinical trials for Midwest Research Group. He is also a founding partner of St. Charles Psychiatric Associates, where he treats adults, adolescents, and children with ADHD. Dr Greg Mattingly is board-certified in adult and adolescent psychiatry and has been a principal investigator in over 300 clinical trials focusing on ADHD and related conditions.
Having served on numerous national and international advisory panels, Dr Mattingly has received awards and distinctions for clinical leadership and neuroscience research. He currently serves on the board of directors for the American Professional Society of ADHD and Related Disorders (APSARD).
Dr Mattingly was paid as a consultant for Takeda and Lundbeck.
No significant effect on body weight in 6 short‑term MDD studies and 1 long‑term MDD study1
Change in weight: randomized, double-blind, placebo-controlled studies1,11

No statistical analyses were conducted with regard to weight change in the US‑based long-term study (Study 10); weight increase was a reported adverse reaction in the TRINTELLIX 20 mg group (≥5%).11
Some reports of weight gain have been received since product approval1
Short-term: TRINTELLIX 5 mg/day to 20 mg/day had no significant effect on body weight as measured by the mean change from baseline in 6- to 8-week placebo‑controlled studies1
Long-term: In the 6-month, double-blind, placebo-controlled phase of the non-US long-term study (Study 9) in patients who had responded to TRINTELLIX during the initial 12-week, open-label phase, there was no significant effect on body weight between TRINTELLIX and placebo-treated patients1

Switching from SSRIs to TRINTELLIX resulted in statistically superior improvement in TESD vs escitalopram in MDD patients1,15
Head-to-head study results showed switching to TRINTELLIX lessened SSRI-induced sexual side effects more than switching to escitalopram in adult MDD patients.1
Mean change from baseline in CSFQ-14 total score at Week 81,15
Study 11: improvement in TESD1,15
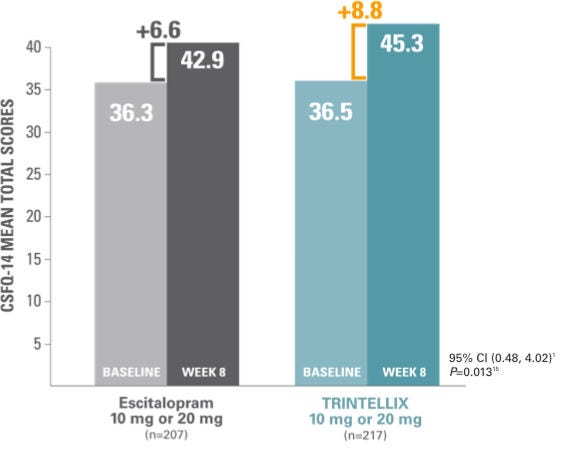
At Week 8, improvement in SSRI‑induced sexual dysfunction in MDD patients switched to TRINTELLIX was superior to improvement in patients who switched to escitalopram.
Please see Warning and Precaution for Sexual Dysfunction listed in the Important Safety Information below
A randomized, double-blind, 8-week study compared TRINTELLIX (n=217) with escitalopram (n=207).
- Adult patients with MDD who were being effectively treated but experiencing SSRI-induced sexual dysfunction (from citalopram, paroxetine, or sertraline) switched to either TRINTELLIX or escitalopram
- Both groups started on 10 mg/day then increased to 20 mg/day at Week 1, followed by flexible dosing
- Clinically meaningful improvement considered to be 2- to 3-point increase in CSFQ-14 total score1
- The CSFQ-14 is a validated scale that measures sexual function, including the 3 phases of the sexual response cycle: desire, arousal, and orgasm
Abbreviations: CI, confidence interval; CSFQ-14, Changes in Sexual Functioning Questionnaire; SNRI, serotonin-norepinephrine reuptake inhibitor; SSRI, selective serotonin reuptake inhibitor; TESD, treatment-emergent sexual dysfunction.
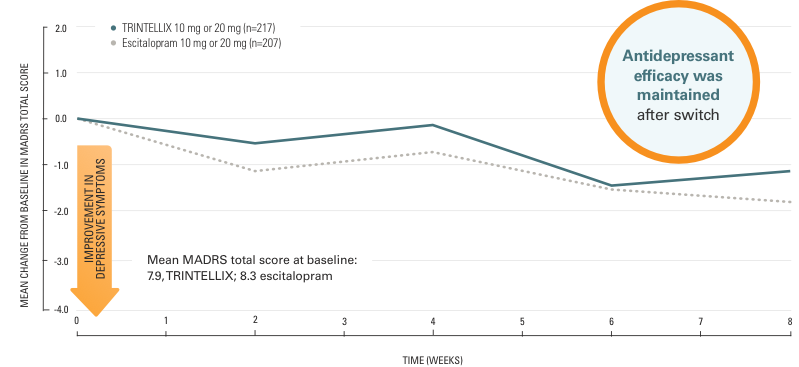
There was no loss in antidepressant effect after switching to TRINTELLIX or escitalopram1
Mean change from baseline in MADRS total score15
- Common adverse events (incidence ≥5% for TRINTELLIX) were nausea (25.0%, 5.4%), headache (9.4%, 7.7%), dizziness (8.0%, 5.0%), and pruritus, generalized (5.8%, 0%) for TRINTELLIX and escitalopram, respectively.15
- Three TRINTELLIX patients experienced a total of 5 SAEs, and 1 escitalopram patient reported 1 SAE.15
- There was 1 case of suicidal behavior in the TRINTELLIX group but no attempted or completed suicides.15
- Twenty TRINTELLIX patients (8.9%) and 14 escitalopram patients (6.3%) withdrew because of an AE.15
- In clinical studies, TESD was both voluntarily and prospectively assessed in patients taking TRINTELLIX.1
- Warning and Precaution for Sexual Dysfunction: Use of serotonergic antidepressants, including TRINTELLIX, may cause symptoms of sexual dysfunction. In male patients, serotonergic antidepressant use may result in ejaculatory delay or failure, decreased libido, and erectile dysfunction. In female patients, use may result in decreased libido and delayed or absent orgasm1

Adverse sexual reactions were also voluntarily and prospectively assessed1
≤5%of female and male patients on TRINTELLIX (vs ≤2% of patients taking placebo) voluntarily reported TESD in 6- to 8-week placebo-controlled studies1†
Voluntary reports of adverse sexual reactions are known to be underreported.
†Voluntarily reported adverse reactions related to sexual dysfunction were captured as individual event terms and aggregated to report overall incidence.1
Prospectively assessed incidence of adverse sexual reactions as reported using the ASEX in patients without sexual dysfunction at baseline in 7 placebo-controlled studies of TRINTELLIX 5 mg/day to 20 mg/day1‡. A separate, self-rated questionnaire was provided to patients during clinical studies.
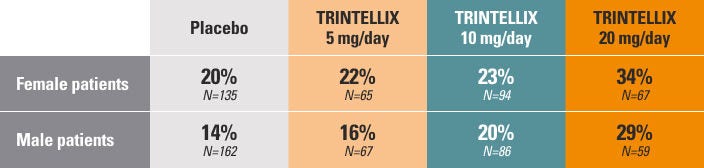
‡Incidence based on the number of subjects with sexual dysfunction during the study/number of subjects without sexual dysfunction at baseline (approximately 1/3 of the population across all study groups).¹ Sexual dysfunction was defined as a subject scoring any of the following on the ASEX at 2 consecutive visits during the study: a total score ≥19; any single item with a score ≥5; or 3 or more items that each had a score ≥4.
Abbreviations: ASEX, Arizona Sexual Experiences Scale; TESD, treatment-emergent sexual dysfunction.
What to explore next?
Long-term
clinical data
Results from multiple clinical studies of TRINTELLIX.

Request a
rep visit
Connect with a TRINTELLIX representative.

Resources for
you and your
patients
Find a range of information, education, and patient resources, as well as industry insights.
Changes in sexual functioning questionnaire short form (CSFQ-14)1
The CSFQ-14 is a validated scale that measures sexual functioning, including the 3 phases of the sexual response cycle:
- Desire
- Arousal
- Orgasm